Relapsed/Refractory B-cell Malignancy Management: Challenges and Advances in Hematology Diseases like Chronic Lymphocytic Leukemia, Mantle Cell Lymphoma, and Waldenström Macroglobulinemia can follow heterogeneous clinical courses in which some patients have disease relapse or resistance following initial therapy.
In this context, targeted therapies have been investigated within the panoply of treatment options. Of these, the zanubrutinib use has been studied investigationally in patients with relapsed or refractory disease. Here we review research insights, clinical considerations and the role of targeted therapy approaches in this setting.
Relapsed disease is when cancer comes back after treatment, and refractory disease is when treatment doesn't work. These are difficult in their own way, and they often challenge us to reconfigure how we handle them.
B-cell malignancies show relapse and refractoriness subjected to:
These permutations create a multidimensional map of decision-making, and highlight the importance of further exploration of treatment choices that include later-line use for zanubrutinib.
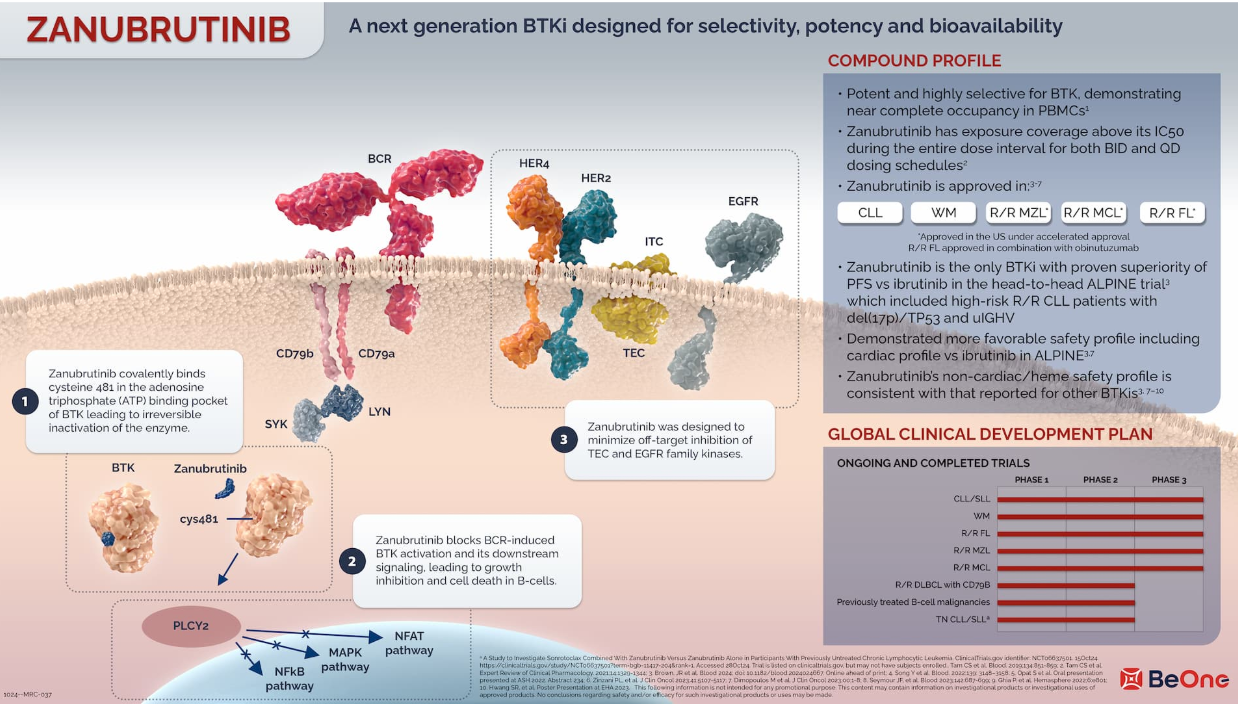
Molecular biology advancements have identified central signalling pathways that mediate malignant B cell survival and proliferation. One obvious connection between the two is Bruton’s Tyrosine Kinase, an enzyme that forms part of B-cell receptor signaling.
Zanubrutinib is an oral BTK inhibitor that comes in capsule form. It was made to only bind to this enzyme.This also interferes with BTK activity, thereby hindering downstream signaling systems involved in B-cell features.
This mechanism underlies the study of zanubrutinib in patients whose disease has relapsed or refractory to prior therapies.
Clinical studies on targeted therapies in relapsed or refractory populations generally enroll patients with varied treatment histories. These studies might include patients who have received multiple prior therapies, including chemotherapy, immunotherapy, or other targeted medications.
In these academic settings, the use of zanubrutinib has been evaluated in Phase II and Phase III trials with endpoints defined to measure clinical activity and safety across broad patient populations.
Commonly evaluated endpoints include:
Interpretation of these endpoints must take into account the design of the studies, characteristics of patients and prior therapy exposure.
The highly heterogeneous patient population is a hallmark of relapsed or refractory disease. Patients can vary greatly with respect to:
Zanubrutinib use has been shown to be effective in several clinical settings, but responses may vary not only by disease type but also based on molecular characteristics and treatment history.
Thus, treatment decisions in relapsed or refractory settings are by definition complex. You can have a conversation in the central neural level with more than only an ailment disorder, and you can convey the present disease state as well as the treatment history and general state of welfare for that patient.
Key considerations may include:
In this program, zanubrutinib utilization is reviewed in the context of an overall treatment strategy rather than as a siloed option.
New research still sheds light on the responses to specific therapies in relapsed or refractory B-cell malignancies. Many studies examining zanubrutinib use utilize the drug in various disease subtypes and patient populations.
These investigations may include:
Such data help to advance a body of evidence that is used to deepen clinical understanding, albeit with an ever evolving nature and subject to ongoing assessment in conjunction with formal regulatory review.
Safety evaluation is especially relevant in patients who have received several lines of treatment. Some examples among the clinical studies with Zanubrutinib include: structured monitoring of
These safety signals are evaluated against clinical outcomes to characterize therapy utilization in this unique patient population.
New data are emerging in the treatment of relapsed or refractory B-cell malignancies. Targeted therapies and molecular diagnostics have improved treatment options.
In view of this evolving landscape, the use of zanubrutinib is considered in comparison with:
We are witnessing a shift towards more individualised medicine in haematologic oncology because of these factors.
Beyond clinical trial data, real-world observations provide additional insights on therapies in general-use practice. Patients treated outside clinical trials may exhibit more extensive clinical variability,
Examples of related real-world data pertaining to zanubrutinib may include:
These findings add to what we know about therapy use and are in line with clinical trial results.
Research in the area of relapsed or refractory B-cell malignancies continues to confront pressing clinical questions. Zanubrutinib is also being actively investigated in the following settings:
This is intended to enhance clinical understanding and enable data-informed choices in challenging treatment settings.
Chronic lymphocytic leukaemia and relapsed or refractory B-cell malignancies remain clinical challenges, highlighting the need for consideration of disease biology, prior therapy exposure, and patient-specific factors. Chronic Lymphocytic Leukemia, Mantle Cell Lymphoma, and Waldenström Macroglobulinemia are other often evolving treatment paths as new research is published.
Clinical studies have evaluated antitumor efficacy of zanubrutinib use in these patient populations according to its mechanism of action, which is Bruton's Tyrosine Kinase inhibition. Over time we will learn more as both laboratory and clinical data add to the understanding of targeted therapy strategies in hematologic malignancies.
Author: Prof. Gevorg Tamamyan, MD, MSc, DSc – Professor and Chairman of the Department of Pediatric Oncology and Hematology at Yerevan State Medical University, and CEO of the Immune Oncology Research Institute. He is actively involved in international oncology initiatives and global pediatric cancer research collaborations.
Medical Review: At the time of writing, this article has been medically reviewed by a board-certified hematologist (MD – USA) with expertise in B-cell malignancies and targeted therapy research.